What is SmartFocus® nTMS?
Nexstim NBS 6 with SmartFocus® nTMS technology uses sophisticated navigation tools to visualize the location, orientation and magnitude of the maximum stimulating electric field (E-field) induced when the TMS coil is activated. SmartFocus® nTMS takes into account the unique shape and conductivity of each patient’s brain and the positioning of the coil and uses this information to determine the location and orientation of the maximum induced E-field in the brain.
In addition, advanced algorithms enable the stimulation dose to be quickly and accurately determined for each patient using their own neurophysiological readings. This all makes Nexstim nTMS a truly personalized and accurate nTMS therapy.
SmartFocus® nTMS treatment offers the patient:
- A measured and individualized dose of TMS
- TMS with the accuracy demanded by brain surgeons
- Treatment sessions personalized for the patient and his or her brain
- Assurance the patient receives his or her prescribed dose every time
- Unsurpassed safety and comfort
For clinicians, SmartFocus nTMS supports:
- Real‑time visualization of coil position relative to individual anatomy
- Consistent alignment with the planned stimulation site
- Reproducible set-up and delivery across sessions
- A navigated workflow designed to enhance confidence in protocol execution
Why Nexstim E-Field Navigated TMS is Different?
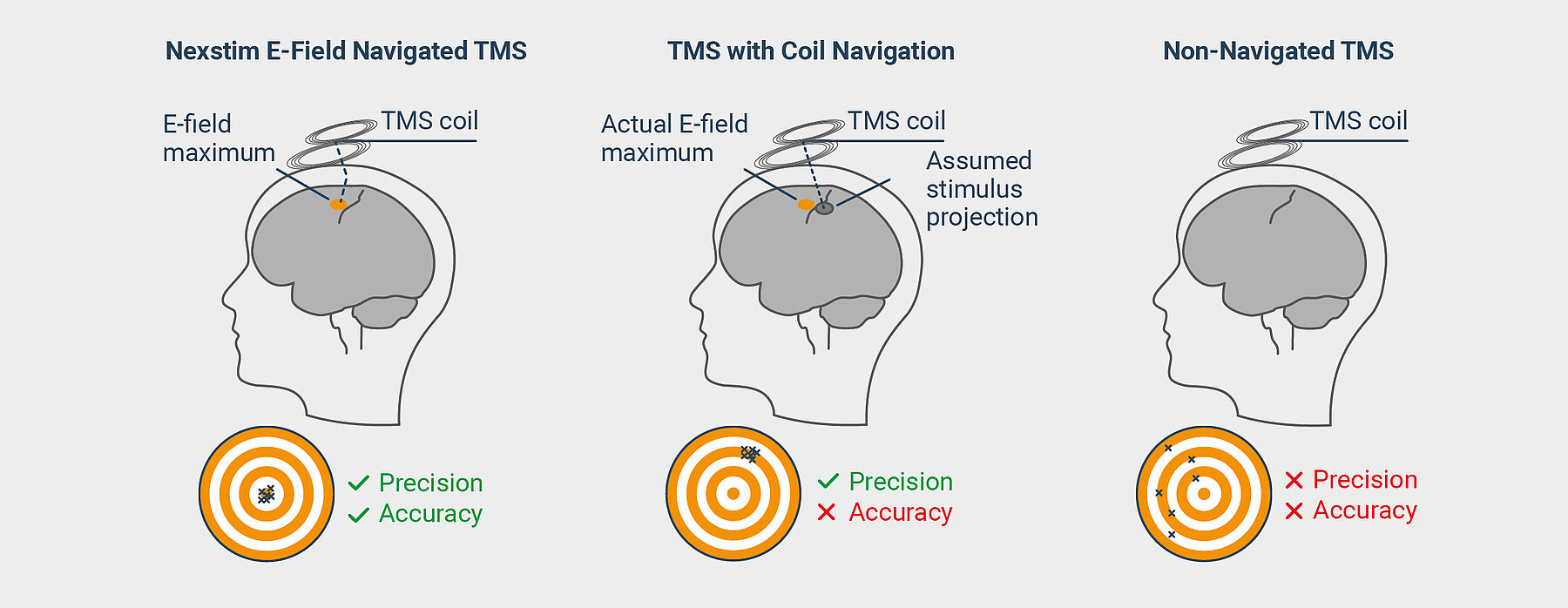
Coil navigation assumes that the stimulation projects perpendicularly from the coil. It does not account for the refraction of the stimulating electromagnetic field caused by bone and brain matter. Nexstim’s E-field navigation takes that refraction into account. Only through visualization of the stimulating E-field it is possible to achieve the precision and accuracy necessary to confirm the right dose is being delivered to the right location.
Yes, I would like to know more
Leave us your contact details, so we can let you know about any upcoming webinars, events in your region or educational resources on nTMS (~6 messages/year). You can unsubscribe from these notifications at any time.
If you would like to see a demonstration of Nexstim nTMS, just add the word “demo” to “further questions and requests”.
Frequently Asked Questions about opening an nTMS clinic
Here are some answers to frequently asked questions we receive from physicians looking to open an nTMS clinic.
Regulatory Approval
Nexstim is a medical device manufacturer. Nexstim NBS 6 is an FDA-cleared and CE-marked medical device (notified body number: 0537). For more information on individual regional clearances, please refer to the table, intended purpose and indications for use descriptions below:
| Clinical Application | European Union (CE mark) | United States (FDA) |
| Pre‑procedural Mapping | CE‑marked | FDA cleared |
| Post‑operative Motor Rehabilitation | CE‑marked | Investigational use only |
| Major Depressive Disorder (Adults) | CE‑marked | FDA cleared |
| Major Depressive Disorder (Adolescents) | Not CE‑marked | FDA cleared |
| Obsessive‑Compulsive Disorder | Not CE‑marked | FDA cleared |
| Chronic Neuropathic Pain | CE‑marked | Investigational use only |
Indications for use & Intended purpose
Pre-procedural mapping (CE mark, FDA clearance, for information on other regional clearances contact Nexstim):
Intended purpose: NBS 6 is intended to be used for localization and assessment of the motor cortex and motor tract integrity for pre-procedural planning purposes. NBS 6 is intended to be used for localization and assessment of cortical areas of speech function for pre-procedural planning purposes.
Indications for use: NBS 6 is indicated for noninvasive mapping of the primary motor cortex of the brain to its cortical gyrus. NBS 6 provides information that may be used in the assessment of the primary motor cortex for pre-procedural planning. NBS 6 is indicated for noninvasive localizations of cortical areas that do not contain essential speech function. NBS 6 provides information that may be used in pre-surgical planning in patients undergoing brain surgery. Intraoperatively, the localization information provided by NBS 6 is intended to be verified by direct cortical stimulation. NBS 6 is not intended to be used during a surgical procedure. NBS 6 is intended to be used by trained clinical professionals.
Post-operative Rehabilitation (CE mark, for information on other regional clearances contact Nexstim):
Intended purpose: NBS 6 is intended to be used for the treatment of surgically induced new or worsening post-operative motor deficits of the upper limb as an adjunct therapy for motor rehabilitation in adult patients having undergone brain tumor surgery. NBS 6 is intended to be used by trained clinical professionals.
Nexstim NBS 6 is not cleared by the FDA for commercial use of post-operative rehabilitation in the United States, for investigational use only.
Chronic Neuropathic Pain (CE mark, for information on other regional clearances contact Nexstim):
Intended purpose: In adult patients suffering from chronic unilateral neuropathic pain, NBS 6 is intended to provide electric field navigated noninvasive repetitive TMS stimulation as therapy to alleviate pain. NBS 6 is intended to be used by trained clinical professionals.
Indications for use: NBS 6 is indicated for MRI-guided and electric field (or E-field) navigated, noninvasive repetitive TMS stimulation (rTMS) of the motor cortex as therapy to alleviate chronic unilateral neuropathic pain in adult patients. Nexstim NBS 6 is intended to be used by trained clinical professionals.
Nexstim NBS 6 is not cleared by the FDA for commercial use of the treatment of chronic pain in the United States, for investigational use only.
Major Depressive Disorder (CE mark, FDA clearance, for information on other regional clearances contact Nexstim):
Intended purpose: NBS 6 is intended to be used for treatment of major depressive disorder (MDD) by targeting and delivering noninvasive repetitive TMS stimulation to the patient's dorsolateral prefrontal cortex.
Indications for use: NBS 6 is indicated for the treatment of Major Depressive Disorder in adult patients who have failed to achieve satisfactory improvement from prior antidepressant medication in the current episode. NBS 6 is intended to be use by trained clinical professionals.
Adolescent Major Depressive Disorder (FDA clearance, for information on other regional clearances contact Nexstim):
Indications for use: NBS 6 is indicated as an adjunct for the treatment of MDD in adolescent patients (age 15-21).
Obsessive-Compulsive Disorder (FDA clearance, for information on other regional clearances contact Nexstim):
Indications for use: NBS 6 is indicated as an adjunct for the treatment of adult patients suffering from obsessive-compulsive disorder (OCD).

