TMS-EEG Methods
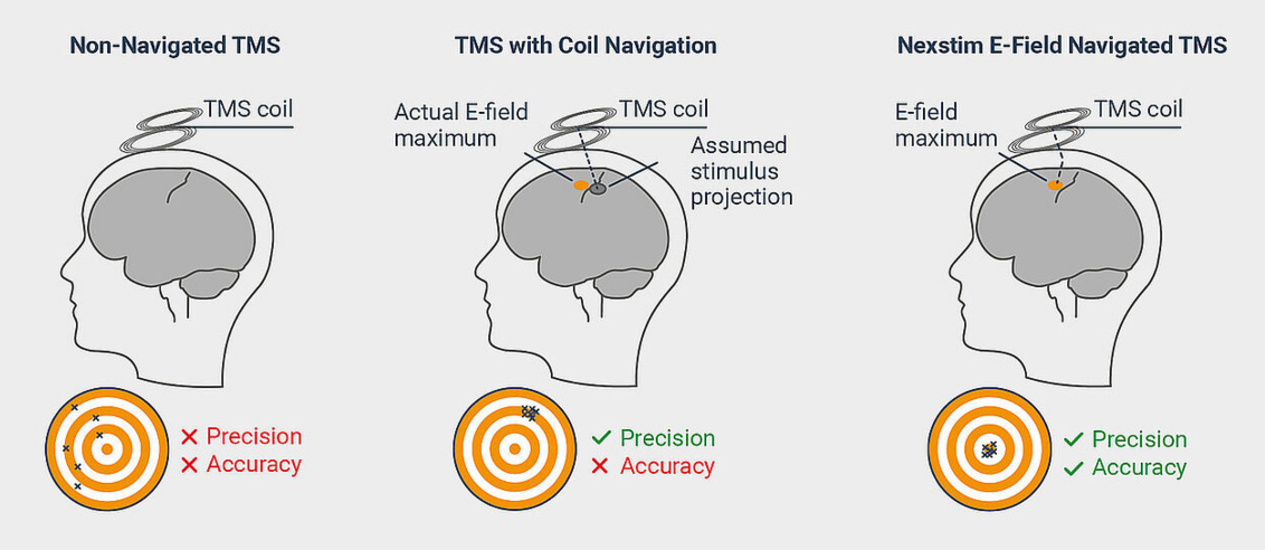
The importance of neuronavigation - repeatability, precision and accuracy
As opposed to standard TMS, where TMS is used without navigation or by simply navigating the coil location, Nexstim integrates advanced real-time electric-field (E-field) modelling to allow the user to visualize the TMS stimulating field with unparalleled accuracy and ease of use.
TMS-evoked EEG responses are sensitive to even the smallest shifts in stimulation location.1 While enabling highly location-specific data to be gathered, this also makes a reliable, precise and accurate navigation solution imperative for TMS-EEG measurements and even more so when studies require repeatability and reproducibility.1,2
As summarized by leading scientists in the field 5:
- Neuronavigation ensures reproducibility of TMS-EEG within and across measurements.
- Neuronavigation with real-time EEG visualization maximizes the TMS cortical impact.
- Neuronavigation is key for reproducible and reliable TMS-EEG cortical mapping.
- Neuronavigation may titrate TMS parameters for TMS-EEG biomarkers and therapies.
- Neuronavigation can guide TMS-EEG by integrating other neuroimaging information.
The importance of the stimulation angle
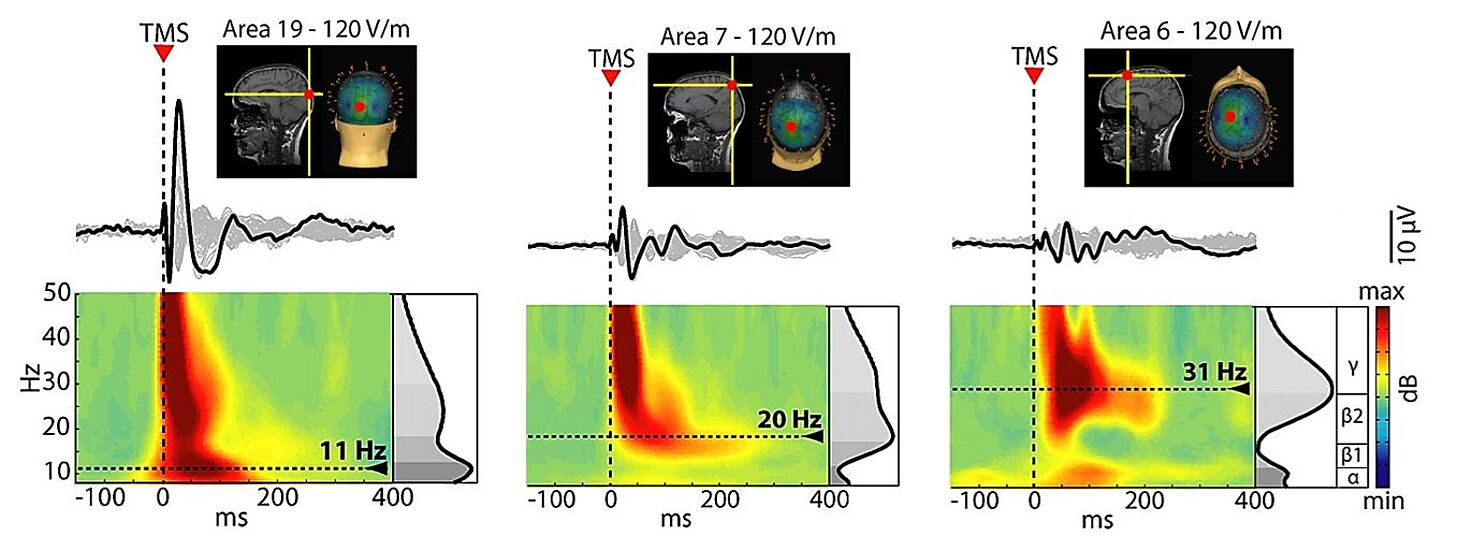
Since the actual impact of TMS on cortical neurons also depends on the orientation of the induced E-field, this figure shows that it is possible to elicit larger early (0-50 ms) EEG responses to TMS by changing the coil orientation while keeping the same target location as well as stimulation intensity. The complete study can be accessed at Casarotto et al. The rt-TEP tool: real-time visualization of TMS-Evoked Potentials to maximize cortical activation and minimize artifacts. J Neurosci Methods. 2022 Mar 15;370:109486.
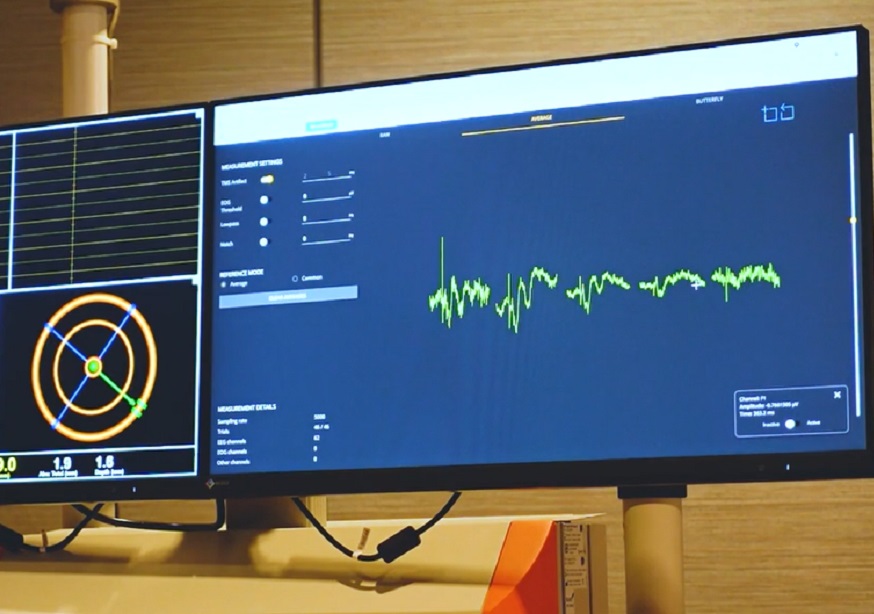
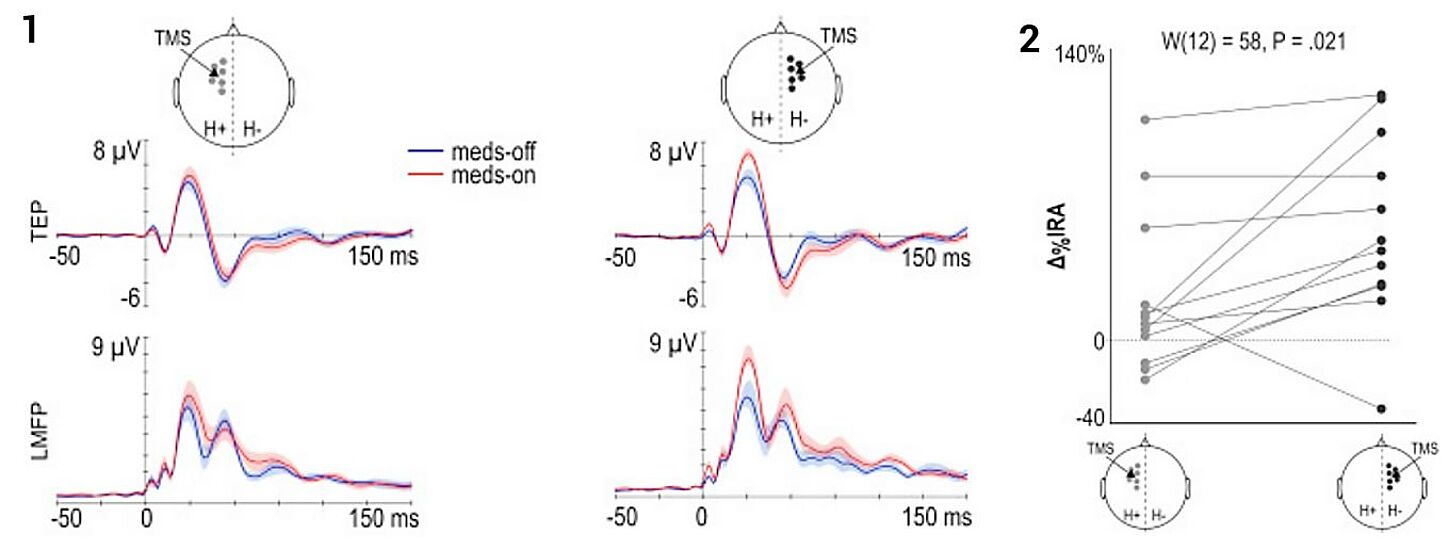
The importance of auditory masking
Panel 2 shows 20-trial average EEG responses to TMS (zoom on frontal channels) when stimulating the left premotor cortex (black cross) at 46% of the maximum stimulator output (MSO). Early (0-50 ms) EEG potentials are low in amplitude (< 4µV) and do not show a clear asymmetry between the two hemispheres. In addition, larger negative-positive deflections are elicited between 100 and 200 ms, with a central distribution. Panel 3 shows how EEG responses to TMS change after increasing stimulation intensity at 55% and after adjusting the noise masking until the subject does not report any auditory perception of the coil's click. In this case, early components (0-50 ms) show larger amplitude (> 10µV), especially in the channels closest to the stimulation target and in the stimulated hemisphere compared to the contralateral one. In addition, slow components with central distribution occurring at latencies between 100 and 200 ms are obliterated. The complete study can be accessed at Casarotto et al. The rt-TEP tool: real-time visualization of TMS-Evoked Potentials to maximize cortical activation and minimize artifacts. J Neurosci Methods. 2022 Mar 15;370:109486.
Dr. Silvia Casarotto, PhD
How to collect and distinguish genuine EEG responses to nTMS
Dr. Silvia Casarotto, PhD, tells in this webinar about how to collect and distinguish genuine EEG responses to nTMS.
- See how to optimize TMS parameters, such as stimulation intensity, based on real-time EEG feedback and reduce signal artifacts.
- Learn pre-processing and post-processing steps that may reduce pulse and muscle artifacts.
Silvia Casarotto, PhD / Sasha D'Ambrosio, PhD / Mario Rosanova, MD, PhD / Simone Russo, MD / Kevin Caulfield / Matteo Fecchio, PhD
TMS-EEG workshop (on-demand recording)
During this workshop, experienced researchers in the field perform a live measurement session with neuronavigated TMS-EEG. Their approach in this workshop represents a strategy for maximizing the impact of TMS on the cortex while minimizing the contribution of artifacts and confounding factors, thus ultimately facilitating the collection of reliable brain responses to direct and non-invasive perturbation of different cortical targets.
- See how to mask the coil's click with the help of a customized noise-masking generator (TAAC - TMS-Adaptable Auditory Control software tool)
- Learn how the quality of TMS-evoked EEG potentials can be effectively assessed during data collection through a dedicated real-time software tool (rt-TEP - real-time TMS-evoked EEG potential)
Other interesting publications
Disorders of Consciousness
Dr. Marcello Massimini, MD
How we can probe changes in cortical circuits with EnTMS-EEG
Dr. Marcello Massimini, MD, tells in this webinar about the use of E-field navigated TMS together with EEG to probe and examine changes in cortical circuits.
See how EnTMS-evoked EEG signals change characteristically under different states of vigilance and brain injury, including coma and stroke
Learn how EnTMS-EEG offers a reliable and repeatable brain-based index of consciousness, independent of sensory processing, executive and motor functions
Understand how EnTMS-EEG overcomes current challenges in assessing the level of consciousness in unresponsive patients and potentially offers a tool to predict functional outcomes and guide intervention
View videos demonstrating the set-up and implementation of EnTMS-EEG
Interesting publications
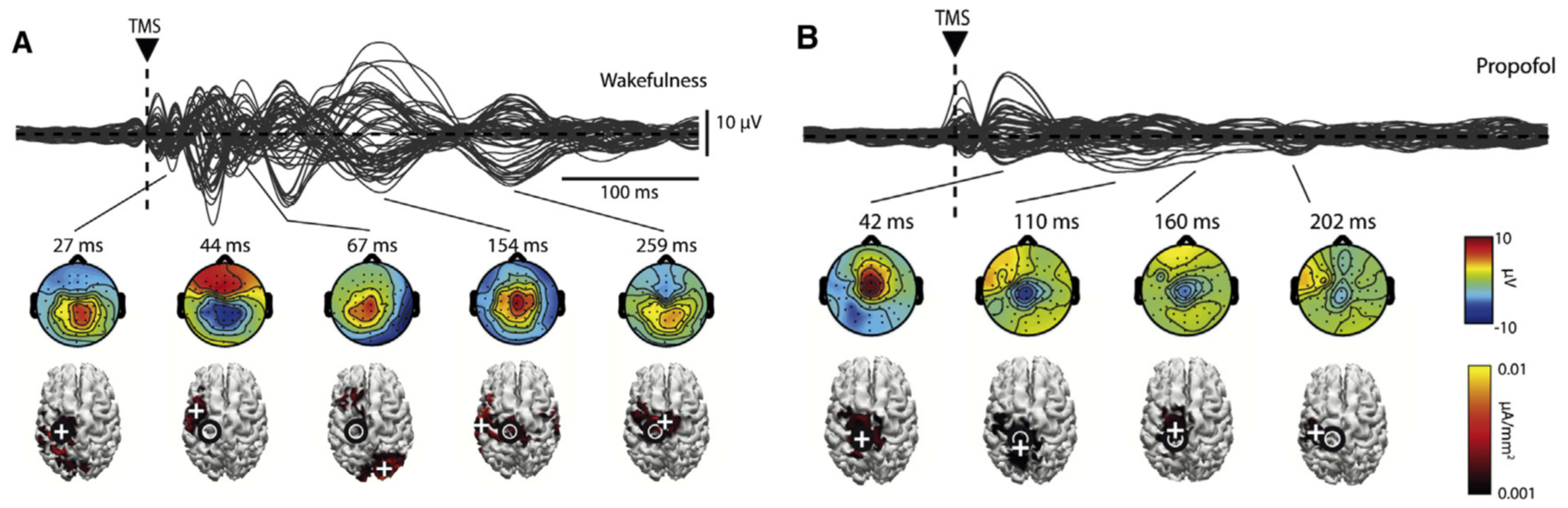
Anesthesia
Mapping stimulus location and orientation
In this study by Sarasso et al. (2015), E-field navigated TMS enabled to map the optimum cortical stimulus location and E-field orientation, to get the strongest TMS-evoked responses. The navigation also allows for the same cortical location to be stimulated accurately, repeatedly for clinical diagnostics.3
Interesting publications
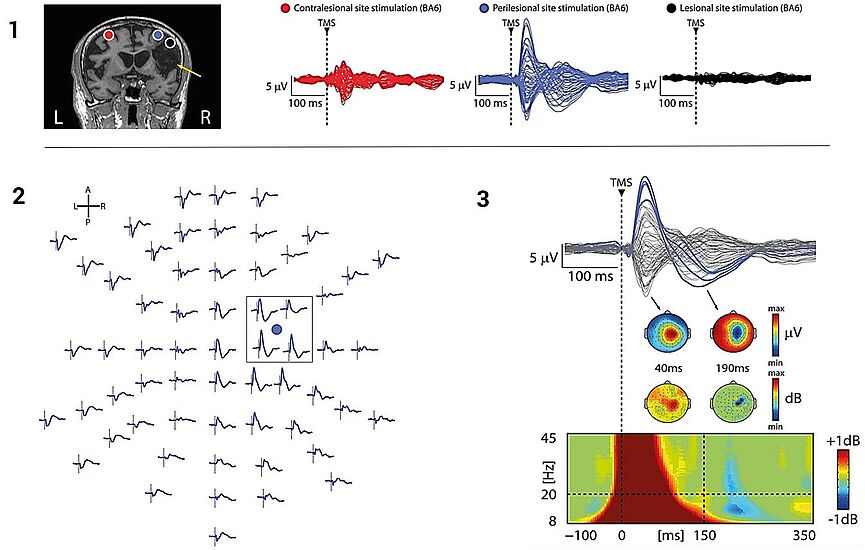
Stroke
Mapping stimulus location and orientation
In this study by Sarasso et al. (2019), E-field navigated TMS enabled to map the optimum cortical stimulus location and E-field orientation, to get the strongest TMS-evoked responses. The navigation also allows for the same cortical location to be stimulated accurately, repeatedly for clinical diagnostics.4
Interesting publications
Alzheimer's
Interview with Paolo M. Rossini on Alzheimer’s disease
Interesting publications
Parkinson's
Cortical excitability changes in the supplementary motor area
Interesting publications
Epilepsy
EEG and rTMS
Researchers at Mayo Clinic - Rochester use EEG and rTMS technologies to bring relief to a man who experienced hundreds of seizures daily:
Interesting publications
Psychiatry
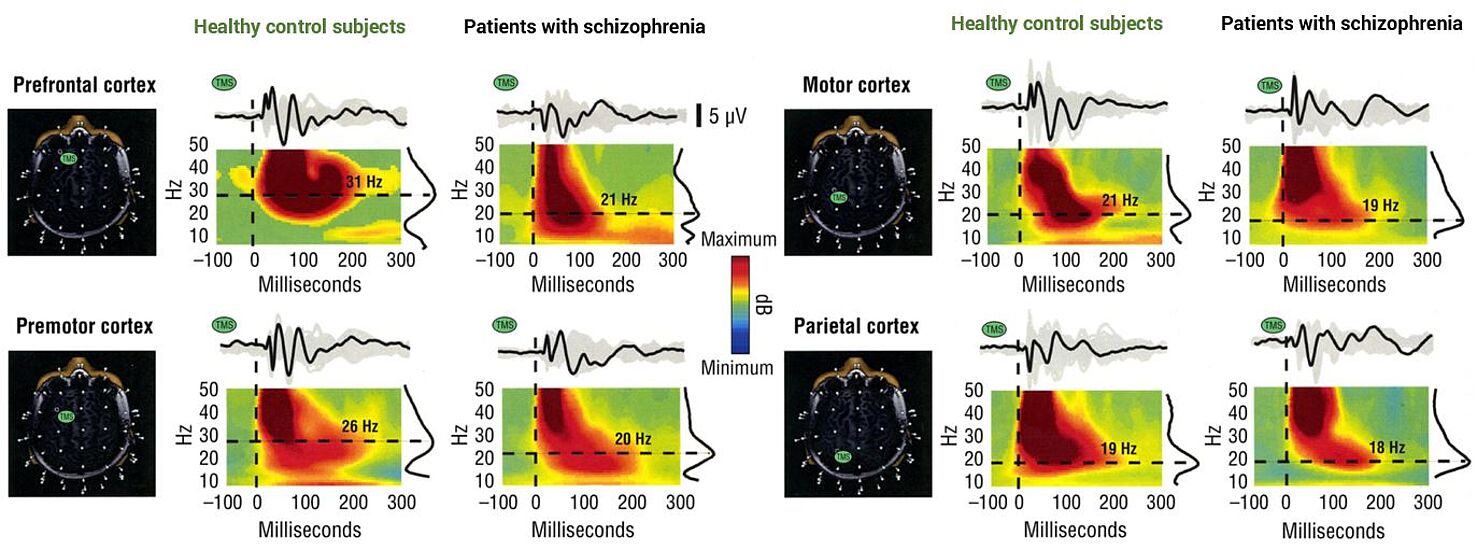
Patients with schizophrenia and TMS
Interesting publications
Neuroscience
Interesting publications
Neurosurgery
TMS-EEG research in neurosurgery
Stay up-to-date on nTMS-EEG
If you leave us your contact details, we are happy to let you know about any upcoming workshops, webinars or events in your region or educational resources on neuronavigated TMS-EEG or nTMS in general (depending on how you select your interests). You can unsubscribe from notifications at any time.
Interested in doing neuronavigated TMS-EEG experiments yourself?

1 Optimize TEP responses
Real-time observation of TMS-evoked potentials is crucial for TMS-EEG experiments.5 Benefit from neuronavigation combined with NBS 6-compatible EEG to help you adjust coil location, orientation and tilt, to optimize the TEP response.
2 Get reproducible results
Reproducible TMS-EEG experiments require consistent TMS stimulation.2 Ensure easily repeatable results with Nexstim’s proprietary, integrated neuronavigation.
3 Minimize artifacts
Minimizing TMS-related artifacts is crucial for TMS-EEG.5 Accurate neuronavigation and optimal coil design allows the activation of cortical points of interest with minimal absolute stimulation intensity, reducing the amplitudes of TMS-pulse artifacts. Furthermore, the Nexstim system has delayed recharging, helping to avoid recharge artifacts.
4 Enhance your targeting
Optional: Utilize fMRI and other functional data overlays in neuronavigation to guide your targeting.
Learn more about our TMS-EEG development projects:
Interested in a live demo of nTMS?
Our team of physicians, researchers, and engineers is prepared to answer your questions. If you would like to learn more or set up a virtual demonstration for your team, please contact us at info@nexstim.com
References
1. Casarotto, S. et al. EEG responses to TMS are sensitive to changes in the perturbation parameters and repeatable over time. PLoS One 5, e10281 (2010).
2. Lioumis, P. et al. Reproducibility of TMS‑evoked EEG responses. Hum. Brain Mapp. 30, 1387–1396 (2009).
3. Sarasso, S. et al. Consciousness and complexity during unresponsiveness induced by propofol, xenon and ketamine. Curr. Biol. 25, 3099–3105 (2015).
4. Sarasso, S. et al. Local sleep‑like cortical reactivity in the awake brain after focal injury. Brain 143, 3672–3684 (2020).
5. Lioumis, P. & Rosanova, M. The role of neuronavigation in TMS–EEG studies: current applications and future perspectives. J. Neurosci. Methods 380, 109707 (2022).