Reported benefits of SmartFocus® nTMS pre-operative mapping:
- With nTMS, interventional strategy and risk can be determined individually and precisely. 1,2
- nTMS maps can help patient better understand the treatment options. 1
- nTMS results can indicate surgery in patients initially deemed inoperable. 1
- In patients with low grade gliomas, increase time before tumor recurrence (progression-free survival time) by 46%. 3
- nTMS maps improve chances for gross total resection rates. 1, 3
- Topographic and neurophysiological preoperative nTMS analysis helps better interpret intraoperatively evoked neurophysiological phenomena. 4
- Patient already prepared and tested for intraoperative language assessment. 5
References
1 Frey D. et al. Navigated transcranial magnetic stimulation improves the treatment outcome in patients with brain tumors in motor eloquent locations. Neuro Oncology 2014: nou110v1-nou110.
2 Frey D. et al. A new approach for corticospinal tract reconstruction based on navigated transcranial stimulation and standardized fractional ansiotrophy values. Neuroimage 2012, 62(3):1600-9
3 Krieg S. et al., Preoperative motor mapping by navigated transcranial magnetic brain stimulation improves outcome for motor eloquent lesions Neuro Oncol. first published online February 9, 2014
4 Krieg S, et al. Utility of presurgical navigated transcranial magnetic brain stimulation for the resection of tumors in eloquent motor areas. J Neurosurg. 2012 May; 116(5):994-1001.
5 Picht T. A comparison of language mapping by preoperative navigated transcranial magnetic stimulation and direct cortical stimulation during awake surgery. Neurosurgery. 2013 May;72(5):808-19.
nTMS makes surgery safer, the resection rates are higher and—in the end—the outcomes are better.
Bernhard Meyer, MD, Professor & Chair,
Department of Neurosurgery, Klinikum rechts der Isar, Technical University of Munich, Germany.
Completing intraoperative monitoring
The data supplied to the FDA compared the agreement between nTMS localization of the primary motor cortex (M1) with M1 localization by direct cortical stimulation (DCS) after craniotomy in the same patients. The data showed excellent agreement between nTMS and DCS localization6.
Prior to surgery, accurate assessment of surgical risk using SmartFocus® nTMS data may modify the indication for intraoperative neurophysiological monitoring (IONM), case-specifically. Robust nTMS maps may be fused with additional imaging modalities on surgical navigation systems to guide surgical resection or optimize the placement of grids and depth electrodes. E-field navigated TMS works synergistically with intraoperative monitoring, expediting DCS motor mapping and facilitating communication between surgeon and clinical neurophysiologist. Topographic and neurophysiological pre-operative nTMS analysis may also facilitate the interpretation of intraoperatively evoked neurophysiological phenomena.
By accurately assessing the surgical risk using nTMS data, the indication for intraoperative neurophysiological mapping and monitoring (IONM) can be made and planned on a case-by-case basis. During surgery, the cortical nTMS data and subcortical nTMS-DTI data help to target and more effectively perform the intraoperative mapping. For example, a phase reversal to determine the central sulcus after prior nTMS mapping can be dispensed with. On the other hand, topographic and neurophysiological pre-operative nTMS analyses help better interpret intraoperatively evoked neurophysiological phenomena.
6 FDA, CDRH Office of Device Evaluation K091457 Dec 2009.
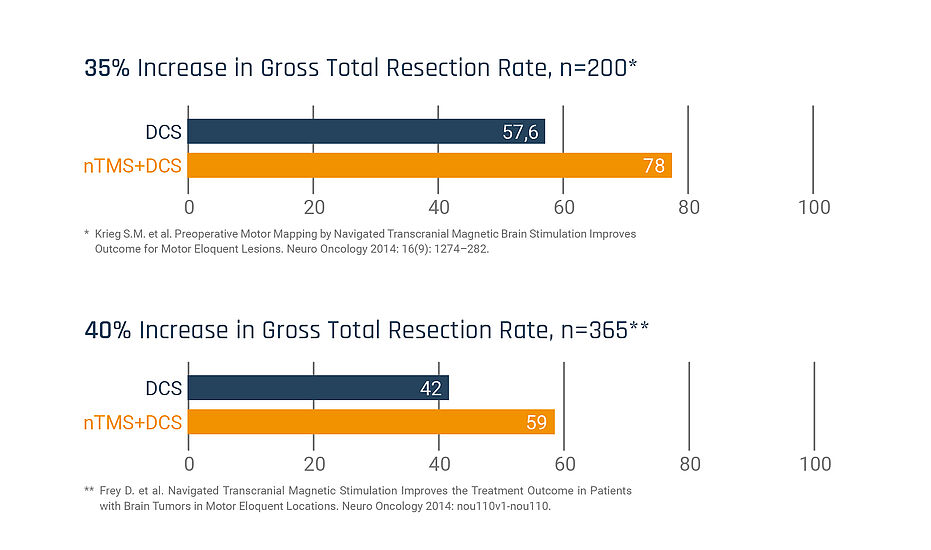
Use of Nexstim NBS SmartFocus® nTMS in motor mapping has increased GTR:
Interested in a live demo of nTMS?
Our team of physicians, researchers, and engineers is prepared to answer your questions. If you would like to learn more or set up a virtual demonstration for your team, please contact us at info@nexstim.com
SmartFocus® nTMS Mapping and Clinical Workflow
Motor Mapping
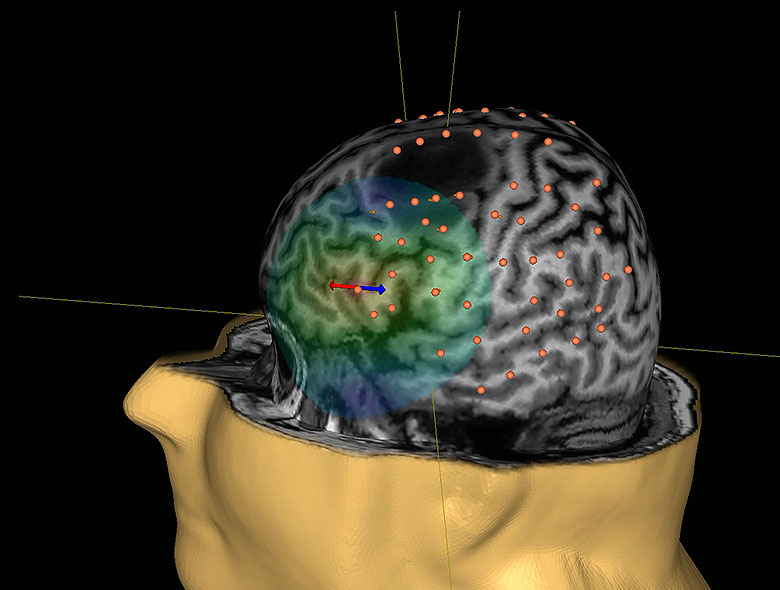
A motor-mapping session typically takes less than an hour. It is important that the patient's muscles are relaxed during the whole session, and they relieve any muscle tension. Neuronavigation of the coil and the head allows the patient to move in the chair.
1. An initial set-up process: The MRI head scan is uploaded to the software. The system creates a 3D model of the patient’s brain and then aligns the coordinate system of that 3D model to a coordinate system you give the patient sitting in the chair.
This alignment process, called registration, is a 2-part process. Part 1 involves showing the system where 3 anatomical landmarks are, both on the MRI (using a digital marker) and on the patient (using a handheld pointer called the digitizing pen). The three recommended anatomical landmarks used in Part 1 of registration are the nasion (bridge of nose) and the left and right crux of helix (specific ripple on outer ear).
Part two of registration involves using the handheld digitizing pen to show the system where specific head surface regions are, when prompted by system.
Once a patient is registered, the infrared camera will track the location of the coil in relation to the head and the user can visualize on the screen the coil and it’s e-field on the 3D model of the brain. You can see where you are stimulating and the system tracks in real time. As you move, you see everything move on the screen.
2. Measuring patients’ resting motor threshold (RMT): EMG (electromyography) surface electrodes are placed on the Abductor Pollicis Brevis (APB) muscles of the patient’s hand. Moving and activating the coil, the operator will find the area in the brain vital for controlling the thumb muscle. Next, the operator will determine how weak the stimulation intensity can be while still causing the thumb muscle to be activated, as can be seen from the EMG tracing displayed on the system screen. Knowing this intensity, the motor threshold, allows the operator to personalize the mapping to be both reliable and comfortable for the patient. Stimulation intensity is nominally set to 110% of RMT.
3. Motor mapping: EMG surface electrodes are placed on the skin over the muscles of interest. These muscles may include the foot, leg and facial muscles, as well as those for the hand, arm and shoulder. The muscles will be chosen based on the lesion's location. The operator will continue to activate the coil, moving it gradually over the head. Normally, only the areas around a tumour are mapped, so the tumour location determines which additional muscles will be monitored.
4. After the mapping: The patient's physician will normally review the results of the SmartFocus® nTMS mapping with the patient. Responses to stimuli will be shown as coloured markers in a 3D model of the brain. Taking into account other diagnostic information, the physician will discuss treatment options. SmartFocus® nTMS results can be of significant help when discussing possible trade-offs between treatment risks and expected benefits.
User-selected responses can be transferred in DICOM format for:
1) DTI-based subcortical fiber tracking,
2) Surgical navigation systems to guide surgical resection or optimize placement of grids and depth electrodes, or
3) Radiosurgery platforms to shift decrease the amount of radiation treating the eloquent tissue.
Mapping language-eloquent cortex
A speech mapping may last 1-1,5 hours in total, but the patient may take breaks, as needed, and the system can be quickly re-aligned by the operator prior to resumption of mapping.
1. An initial set-up process: MRI head scan is uploaded to the software and the patient head is aligned to a 3D rendering of the patient’s MRI head scan created by the software. [see details in the motor mapping part above].
2. Measuring patients’ resting motor threshold (RMT): EMG (electromyography) surface electrodes are placed on the patient’s hand. Moving and activating the coil, the operator will find the area in the brain vital for controlling the thumb muscle. Next, the operator will determine how weak the stimulation intensity can be while still causing the thumb muscle to be activated, as can be seen from the EMG tracing displayed on the system screen. Knowing this intensity, the motor threshold, allows the operator to personalize the mapping to be both reliable and comfortable for the patient. Stimulation intensity is nominally set to 100% of RMT.
3. Testing the Speech-function: Speech function is tested by the patient’s ability to name everyday objects from a series of pictures without stimulation. A subset of familiar objects then forms an initial baseline set, unique for each patient. The object pictures familiar to the patient are re-shown with time-locked delivery of rTMS from the NBS System. Initially, a baseline “language threshold” is established for the individual patient by defining the optimal number of pulses and the frequency of the rTMS pulse train needed to elicit language errors.
4. Language mapping: Using patient-specific thresholds, presumed language areas can then be mapped in a similar manner as in awake surgery. As in intraoperative mapping, language-positive locations are typically defined when speech disturbance occurs in at least two out of three consecutive stimulations.
5. After the mapping: The mapping session is recorded on video. Matching the stimulation locations to speech disturbances allows the neurosurgeon to determine which areas of the brain need to be preserved so that the ability to speak is not harmed. Speech mapping results are a significant help for both patients and doctors when they come to discuss treatment options and any possible trade-offs between risks and expected benefits of surgery.
User-selected responses can be transferred in DICOM format for:
1) DTI-based subcortical fiber tracking,
2) Surgical navigation systems to guide surgical resection or optimize placement of grids and depth electrodes
Yes, I would like to know more
Navigated Transcranial Magnetic Stimulation in Neurosurgery

Find a clinic

Indications for use
The Nexstim Navigated Brain Stimulation (NBS) System 5 is indicated for non-invasive mapping of the primary motor cortex of the brain to its cortical gyrus. The Nexstim NBS System 5 provides information that may be used in the assessment of the primary motor cortex for pre-procedural planning.
Nexstim NexSpeech®, when used together with the NBS System 5, is indicated for non-invasive localization of cortical areas that do not contain essential speech function. NexSpeech® provides information that may be used in pre-surgical planning in patients undergoing brain surgery. Intra-operatively, the localization information provided by NexSpeech® is intended to be verified by direct cortical stimulation.
The Nexstim NBS System 5 and NBS System 5 with NexSpeech® are not intended to be used during a surgical procedure.
The Nexstim NBS System 5 and NBS System 5 with NexSpeech® are intended to be used by trained clinical professionals.
Accuracy of Nexstim NBS System based on comparison with DCS in tumor patients
The accuracy of Nexstim NBS System in clinical use has been evaluated by comparing the localization of cortical representation areas of individual muscles obtained non-invasively by the NBS System in patients with brain tumors to the corresponding locations obtained invasively during surgery by direct cortical stimulation (DCS).
DCS was used as the method of reference as it is considered the gold standard for functional mapping of the motor cortex, and since it provides direct information of the cortical areas connected to peripheral muscles by corticospinal tracts. DCS and NBS both activate cortical neurons with a depolarizing electric field. The DCS electrodes are in direct physical contact with the target cortex surface. The electrode locations can be accurately determined using a neuronavigation system.
A total of 53 muscles in 23 patients were included in the analysis comparing NBS to DCS. In all 23 mpatients the preoperative use of NBS resulted in the localization of the primary motor cortex (M1) to the same gyrus as determined intraoperatively by DCS. In 52 of the 53 muscles (98,1%) the cortical locations eliciting muscle representation areas were located in the same anatomical gyrus by both methods. For the one muscle where the motor areas determined by NBS and DCS were not located in the same gyrus, the area identified by NBS was one gyrus posterior to the area identified by DCS. However, based on the location of all the muscles mapped, also in this patient NBS and DCS results localized the M1 to the same gyrus.
Based on the results of the study, pre-operative NBS localizes the primary motor cortex to the same gyrus as intraoperative DCS. It is recommended that the location of more than one muscle is determined during an NBS examination. Especially in cases where cortical anatomy is unclear or the NBS localization of one or more muscles is unexpected, the stimulation and localization of several muscles as well as the extent of their representation areas in addition to the location of the maximal responses or motor hotspots may provide additional information beneficial for determining the location of motor cortex in relation to the tumour.